Over 15 Years of Successful Clinical Use
PLTMax® is our original, first-generation human platelet lysate developed as an animal serum-free, cell culture supplement and was the first commercially available human platelet lysate.
This product is animal serum-free, however, heparin is added in the manufacturing process of PLTMax® to minimize coagulation. Even with the addition of heparin, clots can spontaneously develop over time. Avoiding long term storage at 4°C can help with reducing the number and size of clots. Since some cell culture media contain high levels of mediators of coagulation like Ca2+ and Mg2+, the addition of heparin is also required when preparing complete media supplemented with PLTMax®.
For PLTMax® production, we use pharmaceutical grade heparin for injection. This heparin is purified and refined from swine according to cGMP methods and is one of the most widely used drugs in humans.
PLTMax® has been a critical ingredient in Phase I, Phase II, and Phase III trials worldwide. It has been used to grow cells for thousands of patients. PLTMax® has a Master File with the FDA to accelerate your application.
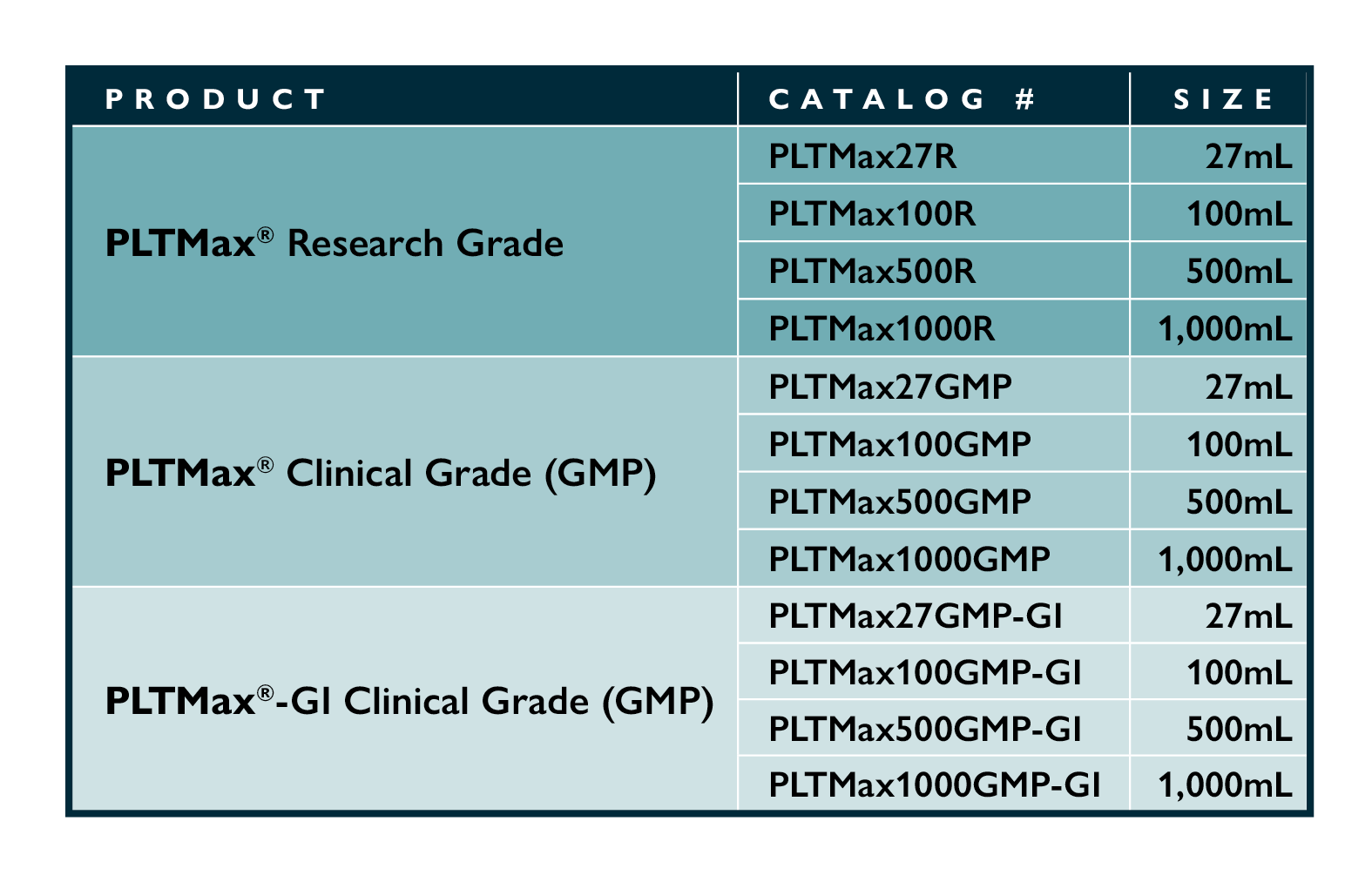
Available in both clinical and research grades in 27mL, 100mL, 500mL, and 1000mL bottles.
Benefits of PLTMax®
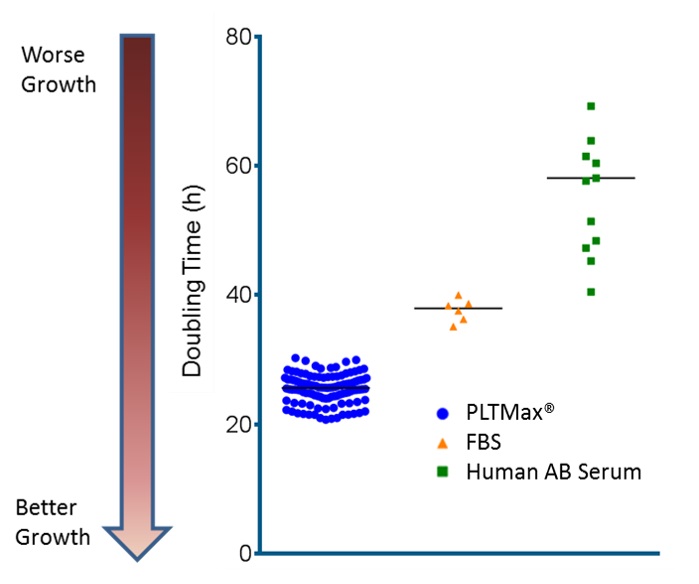
Increased cell growth kinetics (reduced cell doubling time)
Contains growth factors and proteins that maximize cell growth
Enhances genetic stability in MSC cultures
Provides an optimal environment for cell culture and drug discovery
Manufactured to reduce lot-to-lot variation
Not just for MSCs, but also Endothelial cells, T cells, NK cells, tumor cells, iPSCs, and more!
Great for generating cells in large scale bioreactors
Master File with the FDA
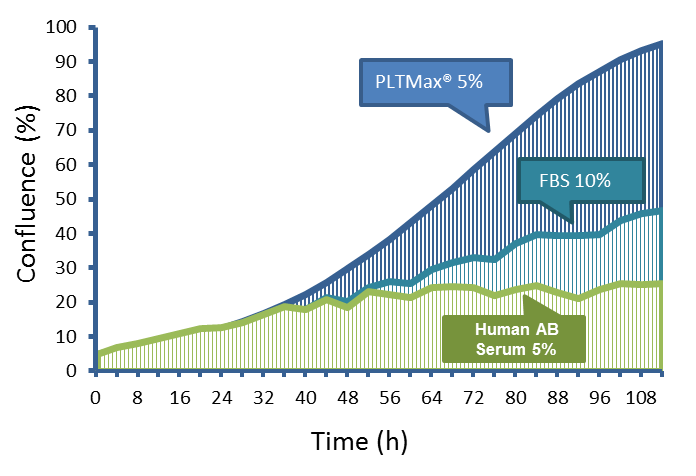
Reduced supplementation (5% or less supplementation with PLTMax® compared to 10% or more FBS and Human AB Serum)
More cost-effective alternative to serum free media supplements
Quality Products to Fit Your Needs
All our products are manufactured under cGMP conditions. We do release testing on all manufactured lots and provide a CoA/CoO for each lot. Some of our release testing includes: pH, osmolality, total protein, mycoplasma, sterility, endotoxin, and a proprietary cell growth assay. We also test a panel of growth factors on all lots for internal information only. Mill Creek is ISO 9001:2015 certified.
Performance of PLTMax® vs Serum Based Products
Time-lapse of Adipose-derived MSCs from day 0 to day 5 of culture
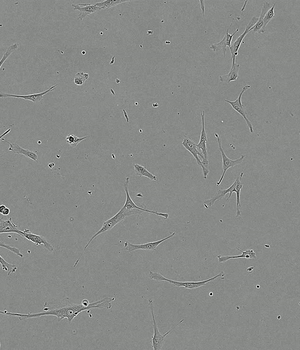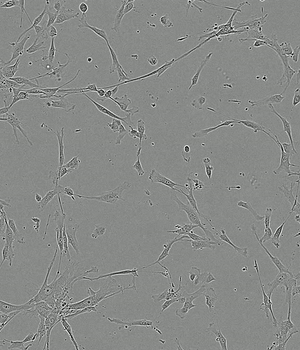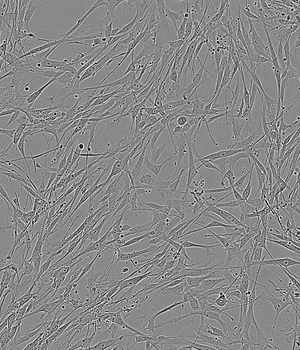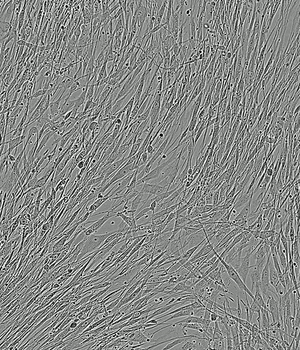
PLTMax® 5%
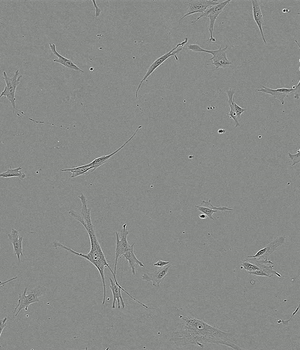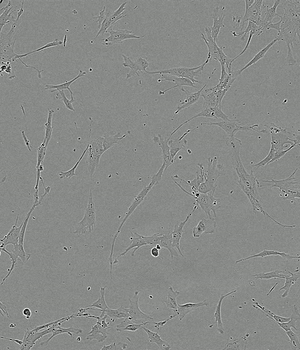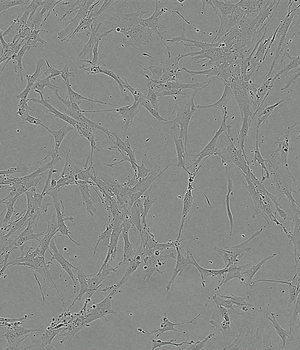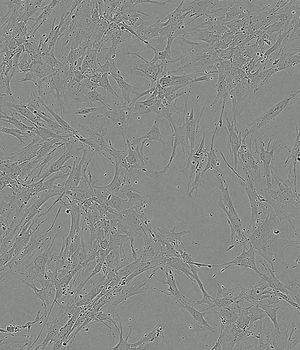
FBS 10%
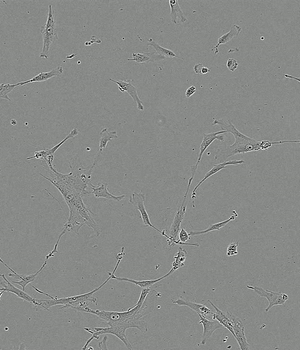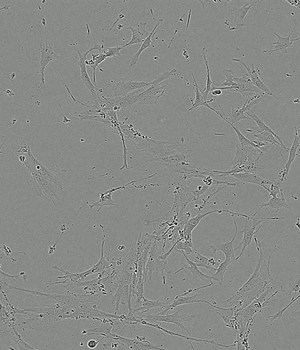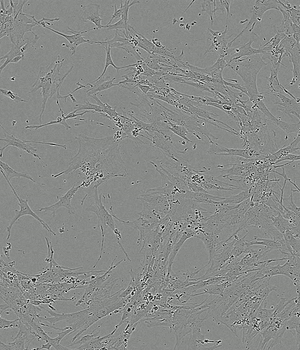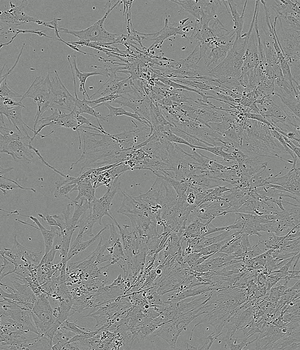
Human AB Serum 5%