Initially developed for use with MSCs, our human platelet lysates (hPLs) have evolved into a platform technology that has been used with successful results with many other cell types and applications, including 2D and suspension cultures, as well as organoids.
Whatever your application may be, our hPL will best feed your growing cell needs!
Some examples of cell types grown with our hPLs are T cells, NK cells, tumor cells, endothelial cells, iPSCs, fibroblasts, hepatocytes and cardiomyocytes.
Mesenchymal Stem Cells (MSCs) are a promising candidate for tissue engineering and regenerative medicine applications due to their capacity of self-renewal and multilineage differentiation, ease of isolation and ability to grow in vitro. Our platelet lysates have demonstrated an outstanding performance expanding MSCs for use in human therapy and are currently being used in hundreds of clinical trials.

Confluence of adipose-derived MSCs expanded for 5 days using our human platelet lysates vs serum derived products.
Chimeric Antigen Receptor (CAR) expressing T cells have demonstrated potent clinical efficacy in patients with hematological malignancies. There are several phase I clinical trials evaluating the use of CAR-T cells for targeting of solid tumor-associated antigens. Some of the challenges found during production of CAR-T cells are the efficiency of T cell transduction to generate CAR-T cells, the expansion of T cells to clinically relevant numbers and the long term survival in vivo of the therapeutic cells. The use of human platelet lysate has demonstrated to aid in the transduction of primary T cells, as well as to improve T cell expansion and prolong in vivo survival of cells. Our platelet lysates, offer an improved performance in T cell expansion versus serum derived products, but also versus other platelet lysates on the market. PLTGold® and PLTGold®-GI efficiently promote T cell expansion, with higher cell yields and lower cell exhaustion rates.
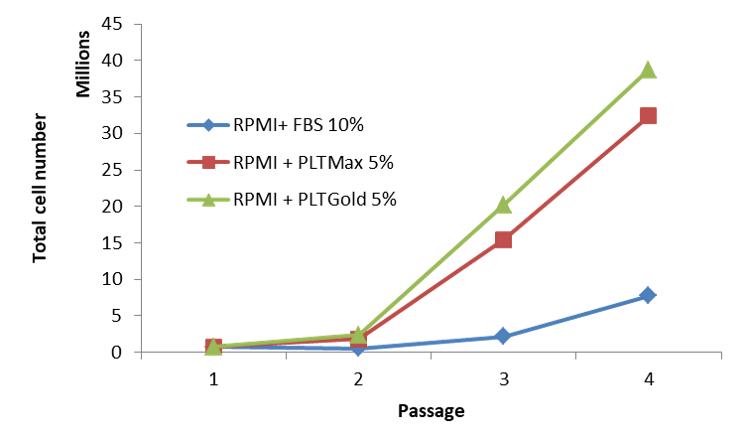
Comparison between our human platelet lysates and fetal bovine serum (FBS) used as supplements for the expansion of T cells.
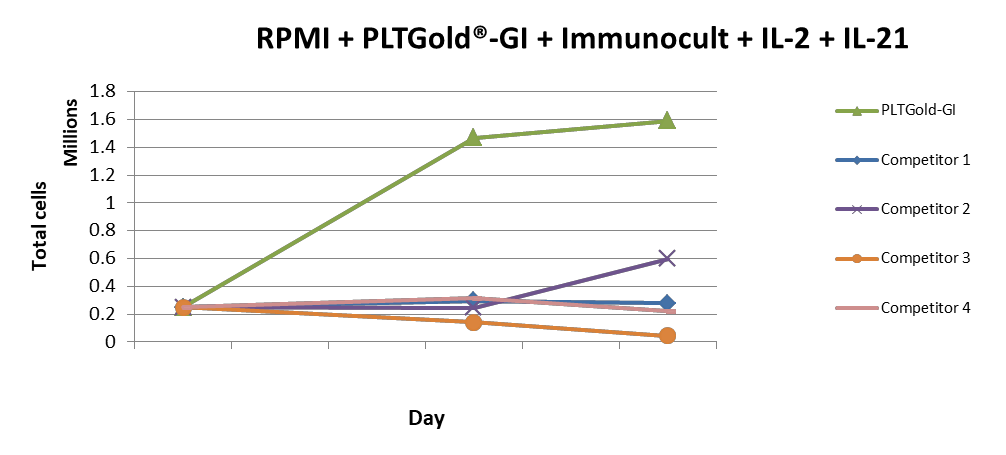
Comparison between PLTGold®-GI and other competitor gamma irradiated platelet lysates used as supplements for the expansion of T cells.
Additionally, our hPLs are suitable for suspension culture of T cells, facilitating the potential large-scale expansion in suspension of allogeneic CAR-T cells.
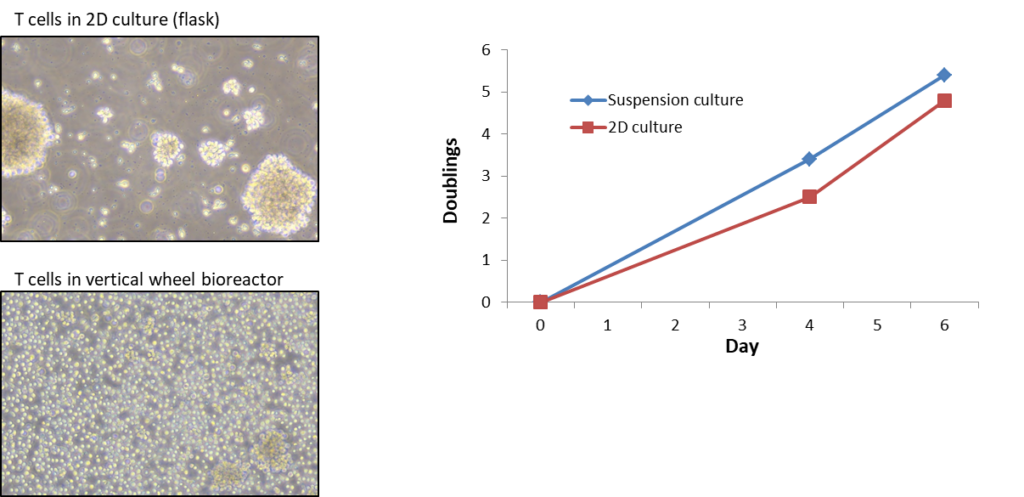
PLTGold®-GI supports T cell growth in suspension. Cell yield obtained when using PLTGold®-GI to grow cells in suspension in bioreactors is better than the yield obtained when cells are grown in flasks in 2D. T cells grown in suspension in vertical wheel bioreactors remain mainly in a single cell suspension for the duration of the culture in comparison to the classic T cell culture look where T cells grow in cell clumps.
All products available on the market to grow endothelial cells contain either FBS or Bovine Brain Extract (BBE), which make them less appropriate, or not appropriate at all, to grow cells that will be used in clinical applications. EndoGoTM XF supplemented with our human platelet lysates, in addition to being animal product free, promote endothelial cell growth in a more efficient way than other products available in the market.
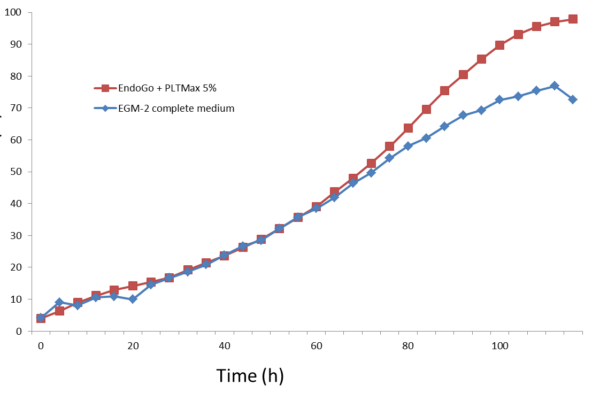
HUVEC grown in EndoGo supplemented with PLTMax® in comparison to the EGM-2 complete medium.
Mill Creek Life Sciences (MCLS) has the license to a patent for the use of platelet lysate to grow human tumor cells. The properties of platelet lysate make it an ideal supplement to efficiently expand primary tumor cells used on research or in translational medicine.
Publications:
MCLS’s PLTMax® has been used successfully in several 3D matrices. Since PLTMax® contains all the clotting factors present in platelets, PLTMax® can additionally be used to generate gels containing high concentrations of growth factors that could potentially be used for 3D culture of cells.
Publications:
